Enlarged lymph nodes in the armpit, medically termed axillary lymphadenopathy, represent a common clinical finding that can cause considerable concern for women. These small, bean-shaped structures serve as crucial components of the immune system, filtering harmful substances and producing infection-fighting cells. When lymph nodes become enlarged, they signal that the body’s immune system is actively responding to various stimuli, ranging from benign infections to more serious underlying conditions.
The axillary region contains multiple lymph node groups that drain lymphatic fluid from the arm, chest wall, and breast tissue. In women, approximately 75% of breast lymphatic drainage flows through these axillary nodes, making their assessment particularly significant in breast health evaluation. Understanding the various causes of axillary lymphadenopathy enables proper recognition of when medical attention is warranted and helps differentiate between conditions requiring immediate intervention and those that may resolve spontaneously.
Clinical presentation can vary significantly, with some enlarged nodes feeling soft and mobile whilst others may appear firm and fixed. The size, consistency, tenderness, and associated symptoms provide valuable diagnostic clues about the underlying aetiology. Early recognition and appropriate evaluation of enlarged axillary lymph nodes can lead to timely diagnosis and treatment of both benign and potentially serious conditions.
Anatomical structure and location of axillary lymph nodes in women
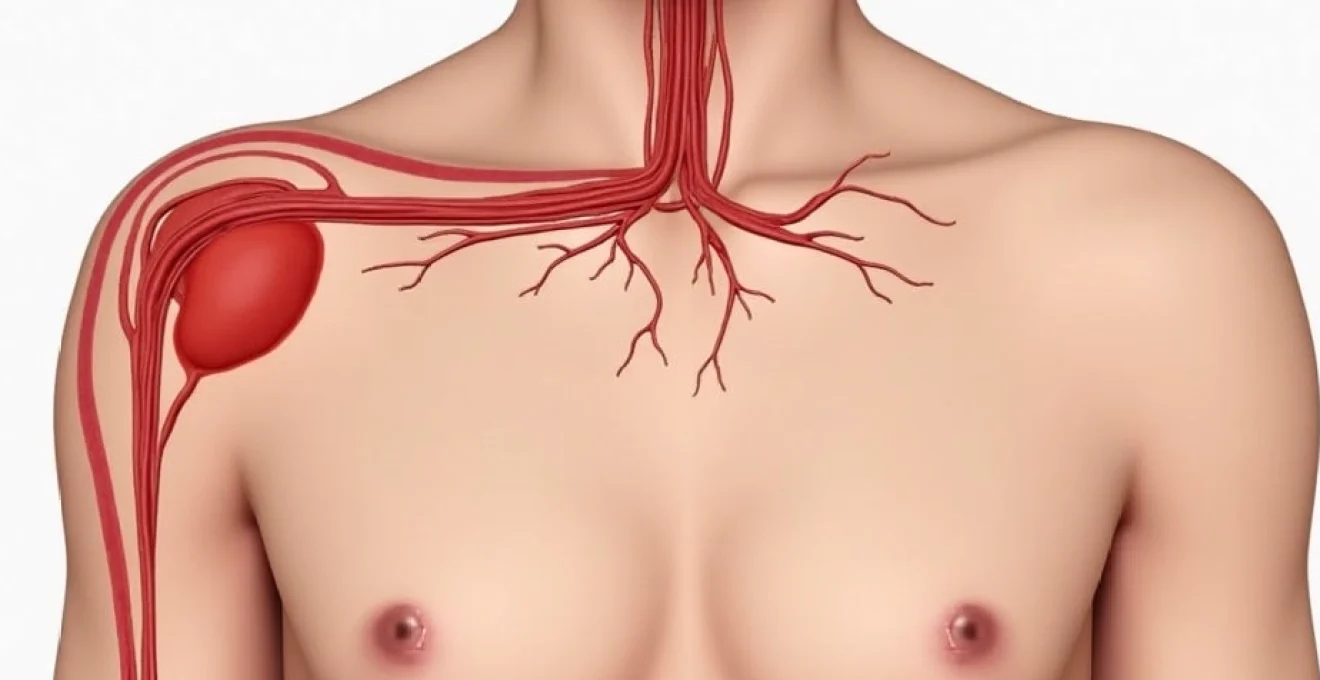
The axillary lymphatic system comprises a complex network of interconnected nodes strategically positioned to filter lymphatic drainage from the upper limb, chest wall, and breast tissue. This intricate arrangement consists of approximately 20 to 40 lymph nodes distributed across five distinct anatomical groups, each serving specific drainage territories. Understanding this anatomical framework proves essential for healthcare professionals conducting clinical examinations and for women performing self-assessments.
Anterior pectoral lymph node group distribution
The anterior pectoral group, also known as the anterior axillary nodes, positions itself along the medial border of the pectoralis minor muscle. These nodes typically number between three to five and receive primary lymphatic drainage from the medial and central portions of the breast, as well as the overlying skin. Their strategic location makes them often the first site of breast cancer metastasis, earning them the designation of sentinel nodes in many clinical scenarios.
During clinical examination, these nodes can be palpated by placing fingers along the anterior axillary fold whilst the patient’s arm remains relaxed at their side. Normal anterior pectoral nodes typically measure less than one centimetre in diameter and feel soft and mobile beneath the skin. When enlarged, they may become more prominent and easier to detect through routine palpation techniques.
Central axillary lymph node positioning
The central axillary group occupies the central portion of the axillary space, embedded within the axillary fat pad. These nodes receive lymphatic drainage from the anterior pectoral, posterior subscapular, and lateral humeral groups, making them a convergence point for much of the upper limb and chest wall lymphatic flow. Their central location renders them readily accessible during clinical examination when properly positioned.
Healthcare professionals can best palpate these nodes by having the patient slightly abduct their arm whilst supporting the limb. The examining fingers should press gently against the central chest wall within the axillary hollow, moving systematically to assess for enlarged or abnormal nodes. Bilateral comparison proves particularly valuable in identifying unilateral abnormalities that may warrant further investigation.
Posterior subscapular lymph node anatomy
The posterior subscapular nodes, positioned along the subscapular vessels near the posterior axillary fold, drain lymphatic fluid from the posterior chest wall and back regions. These nodes typically remain smaller and less numerous than their anterior counterparts but can become significantly enlarged in conditions affecting the posterior thoracic region or in systemic lymphatic disorders.
Examination of the posterior subscapular group requires specific positioning, with the patient’s arm slightly elevated and the examiner’s fingers positioned along the posterior aspect of the axillary space. These nodes may be more challenging to palpate in individuals with increased subcutaneous tissue, requiring careful systematic palpation techniques to ensure thorough assessment.
Lateral humeral lymph node classification
The lateral humeral group, also termed lateral axillary nodes, positions itself along the upper portion of the humerus near the axillary vessels. These nodes primarily drain the lateral aspects of the arm and may become enlarged in response to infections or injuries affecting the upper limb. Their anatomical position makes them less commonly involved in breast-related pathology compared to the anterior pectoral group.
Clinical examination of lateral humeral nodes requires positioning the patient’s arm in slight abduction whilst palpating along the lateral aspect of the axillary space. Systematic approach to examination ensures comprehensive assessment of all lymph node groups, preventing oversight of potentially significant findings that could influence diagnostic considerations and treatment planning.
Infectious aetiologies causing axillary lymphadenopathy
Infectious causes represent the most common aetiology of enlarged axillary lymph nodes in women, accounting for approximately 60-70% of all cases presenting to primary care settings. The immune system’s response to bacterial, viral, or other pathogenic organisms triggers lymph node activation and subsequent enlargement as the nodes work to filter and eliminate infectious agents. These reactive changes typically develop relatively quickly and may be accompanied by systemic symptoms such as fever, malaise, and localised inflammation.
Staphylococcus aureus and streptococcus pyogenes infections
Bacterial infections caused by Staphylococcus aureus and Streptococcus pyogenes frequently result in axillary lymphadenopathy, particularly when the primary infection site involves the arm, hand, or breast tissue. These gram-positive bacteria commonly cause cellulitis, impetigo, or wound infections that trigger regional lymph node enlargement as part of the body’s immune response. The affected nodes typically become tender, warm, and may develop into suppurative lymphadenitis if left untreated.
Staphylococcal infections often present with characteristic features including localised erythema, warmth, and purulent drainage at the primary site. The associated lymphadenopathy usually develops within 24-48 hours of symptom onset and may progress rapidly if appropriate antibiotic therapy is not initiated. Methicillin-resistant Staphylococcus aureus (MRSA) infections require particular attention due to their resistance to standard antibiotic regimens and potential for serious complications.
Streptococcal infections, particularly those caused by Group A Streptococcus, can produce extensive lymphangitis with prominent red streaking extending from the infection site toward the axillary region. This presentation, sometimes called “blood poisoning” by patients, represents active lymphatic spread and requires immediate medical attention to prevent systemic complications.
Cat scratch disease from bartonella henselae
Cat scratch disease, caused by the intracellular bacterium Bartonella henselae, presents as a distinctive syndrome characterised by regional lymphadenopathy following feline contact. The condition typically begins with a small papule or pustule at the inoculation site, usually on the arm or hand, followed by significant enlargement of the draining lymph nodes within one to three weeks. Axillary lymphadenopathy develops when the scratch or bite occurs on the arm or hand.
The enlarged lymph nodes in cat scratch disease often become markedly swollen, sometimes reaching several centimetres in diameter, and may remain enlarged for several weeks to months. Unlike many bacterial infections, the nodes typically remain non-suppurative, though occasional abscess formation can occur. Parinaud’s oculoglandular syndrome represents a specific variant where conjunctival inoculation leads to preauricular rather than axillary lymphadenopathy.
Diagnosis relies heavily on exposure history, with approximately 90% of patients reporting recent contact with cats, particularly kittens. The condition generally follows a benign, self-limiting course, though immunocompromised individuals may develop more severe manifestations requiring antibiotic intervention with agents such as azithromycin or doxycycline.
Mycobacterial infections including tuberculosis lymphadenitis
Mycobacterial infections, including both Mycobacterium tuberculosis and non-tuberculous mycobacteria, can cause chronic axillary lymphadenopathy with distinctive clinical characteristics. Tuberculous lymphadenitis typically presents as painless, gradually enlarging nodes that may become matted together over time. The condition often affects multiple lymph node regions simultaneously and may be accompanied by constitutional symptoms such as night sweats, weight loss, and low-grade fever.
Non-tuberculous mycobacterial infections, particularly those caused by M. avium complex, occur more commonly in immunocompromised patients or those with underlying lung disease. These infections tend to follow a more indolent course than typical bacterial infections and may require prolonged antimicrobial therapy with combinations of macrolides, ethambutol, and rifamycin derivatives.
Interferon-gamma release assays (IGRAs) and tuberculin skin tests provide valuable diagnostic information, though their interpretation requires consideration of the patient’s immune status and previous BCG vaccination history. Tissue biopsy with acid-fast staining and mycobacterial culture remains the definitive diagnostic approach for confirming mycobacterial lymphadenitis.
Viral infections: Epstein-Barr virus and cytomegalovirus
Viral infections commonly cause generalised lymphadenopathy that includes axillary node enlargement, with Epstein-Barr virus (EBV) and cytomegalovirus (CMV) representing two of the most significant aetiologies. EBV infection, the causative agent of infectious mononucleosis, typically affects adolescents and young adults, producing the classic triad of fever, sore throat, and lymphadenopathy. The axillary nodes become enlarged as part of the generalised lymphadenopathy that characterises this condition.
Cytomegalovirus infections can produce similar clinical manifestations, though they tend to be milder and may go unrecognised in immunocompetent individuals. In immunocompromised patients, CMV can cause more severe disease with pronounced lymphadenopathy and systemic complications. Both EBV and CMV infections can produce atypical lymphocytes visible on peripheral blood smears, providing valuable diagnostic clues.
The lymphadenopathy associated with viral infections typically develops gradually over several days to weeks and may persist for months after acute symptom resolution. Supportive care remains the mainstay of treatment for most viral lymphadenitides, though antiviral agents may be considered in severe cases or immunocompromised patients.
Malignant neoplasms associated with axillary lymph node enlargement
Malignant causes of axillary lymphadenopathy represent a critical diagnostic consideration, particularly in women over 40 years of age or those with risk factors for cancer. Neoplastic involvement can occur through direct extension from primary tumours, metastatic spread via lymphatic channels, or primary lymphoid malignancies arising within the nodes themselves. The characteristics of malignant lymphadenopathy often differ from benign causes, with nodes typically feeling firm, fixed, and non-tender on examination.
The presence of hard, immobile axillary lymph nodes in women warrants immediate medical evaluation, as these characteristics strongly suggest malignant involvement and require urgent diagnostic workup including imaging and tissue sampling.
Primary breast carcinoma metastasis patterns
Breast carcinoma represents the most significant malignant cause of axillary lymphadenopathy in women, with nodal involvement serving as a crucial prognostic factor in staging and treatment planning. The lymphatic drainage patterns of the breast direct malignant cells primarily to the anterior pectoral and central axillary node groups, though any axillary node group may be affected depending on the primary tumour location and growth characteristics.
The presence of axillary nodal metastases significantly impacts breast cancer staging, with the number of involved nodes and the extent of metastatic disease influencing both prognosis and treatment recommendations. Current staging systems categorise nodal involvement as N1 (1-3 positive nodes), N2 (4-9 positive nodes), or N3 (10 or more positive nodes), with higher categories associated with decreased survival rates and more aggressive treatment approaches.
Sentinel lymph node biopsy has revolutionised the surgical management of breast cancer, allowing for precise identification of the first lymph node or nodes to receive lymphatic drainage from the primary tumour. This technique enables accurate staging whilst minimising the morbidity associated with complete axillary lymph node dissection in patients with negative sentinel nodes. Blue dye and radioactive tracer injection techniques facilitate intraoperative identification of sentinel nodes with high accuracy rates.
Hodgkin’s lymphoma B-Cell manifestations
Hodgkin’s lymphoma represents a distinct form of lymphoid malignancy that commonly presents with enlarged lymph nodes in multiple regions, including the axillary area. This cancer typically affects younger adults, with a bimodal age distribution showing peaks in the third decade and after age 50. The disease characteristically spreads in a contiguous manner from one lymph node group to adjacent regions, a pattern that distinguishes it from non-Hodgkin’s lymphomas.
The pathognomonic Reed-Sternberg cells serve as the diagnostic hallmark of Hodgkin’s lymphoma, representing malignant B-lymphocytes surrounded by a reactive inflammatory infiltrate. Classical Hodgkin’s lymphoma subtypes include nodular sclerosis, mixed cellularity, lymphocyte-rich, and lymphocyte-depleted variants, each with distinct histological characteristics and clinical behaviours.
Patients with Hodgkin’s lymphoma may experience B-symptoms, including unexplained fever, drenching night sweats, and weight loss exceeding 10% of body weight within six months. Alcohol-induced pain in involved lymph nodes represents an unusual but characteristic symptom that may provide diagnostic clues when present.
Non-hodgkin’s lymphoma diffuse large B-Cell type
Non-Hodgkin’s lymphomas encompass a diverse group of lymphoid malignancies that can present with axillary lymphadenopathy, with diffuse large B-cell lymphoma (DLBCL) representing the most common subtype. Unlike Hodgkin’s lymphoma, non-Hodgkin’s lymphomas tend to spread in a random, non-contiguous pattern and may involve extranodal sites at presentation more frequently.
DLBCL typically presents as rapidly enlarging lymph nodes that may reach considerable size within weeks to months of onset. The aggressive nature of this malignancy necessitates prompt diagnosis and treatment initiation to optimise patient outcomes. The disease commonly affects older adults, with a median age at diagnosis of approximately 65 years, though it can occur at any age.
Staging evaluation for non-Hodgkin’s lymphoma requires comprehensive assessment including computed tomography imaging, bone marrow biopsy, and assessment of lactate dehydrogenase levels. Positron emission tomography (PET) scanning provides valuable information about metabolic activity and treatment response monitoring in many lymphoma subtypes.
Melanoma metastatic spread to regional lymphatics
Melanoma represents a potentially aggressive form of skin cancer that commonly metastasises to regional lymph nodes, including the axillary region when the primary tumour involves the arm, shoulder, or upper back. The propensity for early lymphatic spread makes nodal assessment a critical component of melanoma staging and treatment planning. Sentinel lymph node biopsy has become standard practice for melanomas exceeding 1 mm in thickness or those with high-risk features.
The presence of melanoma cells within axillary lymph nodes dramatically alters the prognosis and treatment approach, often necessitating completion lymph node dissection and consideration of adjuvant systemic therapy. Recent advances in immunotherapy and targeted therapy have significantly improved outcomes for patients with metastatic melanoma, though early detection and treatment remain paramount for optimal results.
Melanoma-involved lymph nodes may present as firm, non-tender masses that gradually increase in size over weeks to months. The nodes may occasionally develop a characteristic blue-black discoloration due to melanin pigment deposition, though this finding is uncommon. Regular self-examination and professional surveillance enable early detection of recurrent or metastatic disease in patients with previous melanoma diagnoses.
Auto
Autoimmune and inflammatory disorders affecting axillary nodes
Autoimmune and inflammatory conditions represent an important category of non-infectious causes of axillary lymphadenopathy in women. These disorders involve dysregulation of the immune system, leading to chronic inflammation that can affect lymph nodes throughout the body, including those in the axillary region. The lymphadenopathy associated with autoimmune conditions typically develops gradually and may wax and wane in correlation with disease activity, distinguishing it from the acute onset seen with infectious causes.
Systemic lupus erythematosus (SLE) commonly causes generalised lymphadenopathy, with axillary nodes becoming enlarged in approximately 50-60% of patients during active disease phases. The nodes typically remain mobile and non-tender, though they may become more prominent during disease flares accompanied by other constitutional symptoms. Antinuclear antibody testing and complement levels provide valuable diagnostic information when SLE is suspected as the underlying cause of lymphadenopathy.
Rheumatoid arthritis can produce reactive lymphadenopathy, particularly in patients with active joint inflammation affecting the upper extremities. The chronic inflammatory state associated with this condition leads to persistent immune system activation, resulting in lymph node enlargement that may fluctuate with disease activity levels. Sjögren’s syndrome, an autoimmune condition affecting salivary and lacrimal glands, can also cause significant lymphadenopathy and carries an increased risk of developing lymphoma over time.
Sarcoidosis represents a multisystem inflammatory disorder that frequently affects lymph nodes, with bilateral hilar lymphadenopathy being the most characteristic finding. However, peripheral lymph nodes, including those in the axillary region, can also become enlarged in approximately 25-30% of patients. The condition typically affects women more frequently than men and may present with associated pulmonary symptoms, skin lesions, or ocular manifestations. Elevated angiotensin-converting enzyme levels and non-caseating granulomas on tissue biopsy support the diagnosis of sarcoidosis when clinical suspicion is high.
Diagnostic imaging modalities for axillary lymphadenopathy assessment
Modern diagnostic imaging plays a crucial role in the evaluation of enlarged axillary lymph nodes, providing detailed anatomical information that guides clinical decision-making and treatment planning. The selection of appropriate imaging modalities depends on the clinical presentation, suspected aetiology, and the need for tissue characterisation or staging information. Each imaging technique offers distinct advantages and limitations that must be considered within the context of the individual patient’s presentation.
Ultrasonography represents the initial imaging modality of choice for evaluating axillary lymphadenopathy due to its non-invasive nature, real-time capability, and excellent soft tissue resolution. High-resolution ultrasound can distinguish between normal and abnormal lymph nodes based on size, shape, echogenicity, and vascular patterns. Normal lymph nodes typically display an oval shape with a hyperechoic hilum, whilst abnormal nodes may appear rounded, hypoechoic, or demonstrate absent hilar architecture.
The use of colour Doppler ultrasound provides additional information about nodal vascularity, with malignant nodes often showing increased peripheral blood flow compared to the central hilar vascularity seen in reactive nodes. Ultrasound-guided fine needle aspiration can be performed simultaneously, allowing for tissue sampling when indicated. The technique demonstrates sensitivity rates of 85-95% for detecting malignant lymphadenopathy when performed by experienced operators.
Computed tomography (CT) scanning offers superior anatomical detail and the ability to assess multiple lymph node regions simultaneously, making it particularly valuable for staging malignant diseases. CT can identify nodes that may not be palpable on clinical examination and provides precise measurements for monitoring treatment response. The use of intravenous contrast enhancement improves tissue characterisation and helps differentiate between inflammatory and malignant processes.
Magnetic resonance imaging (MRI) provides excellent soft tissue contrast and multiplanar imaging capabilities, making it particularly useful for evaluating the relationship between enlarged lymph nodes and surrounding structures. MRI demonstrates superior ability to detect extracapsular nodal extension and can identify subtle changes in nodal architecture that may not be apparent on other imaging modalities. Diffusion-weighted imaging sequences offer functional information about tissue cellularity that can help distinguish between malignant and benign lymphadenopathy.
Positron emission tomography (PET) scanning, often combined with CT (PET-CT), provides metabolic information about lymph node activity that proves invaluable for staging lymphomas and monitoring treatment response. The technique demonstrates high sensitivity for detecting metabolically active disease and can identify involvement in normal-sized lymph nodes that might be missed by anatomical imaging alone. However, inflammatory conditions can also demonstrate increased metabolic activity, potentially leading to false-positive results that require careful clinical correlation.
Clinical examination techniques and palpation methodology for enlarged axillary nodes
Systematic clinical examination of the axillary region requires specific techniques and patient positioning to ensure comprehensive assessment of all lymph node groups. Proper examination technique enables healthcare professionals to detect subtle abnormalities, characterise nodal features, and monitor changes over time. The examination should be performed in a warm, private environment with adequate lighting to ensure patient comfort and optimal assessment conditions.
Patient positioning plays a critical role in successful axillary examination, with the optimal approach involving the patient seated or standing with arms relaxed at their sides. The examiner should position themselves facing the patient, using both hands to perform bilateral simultaneous examination when possible. This technique allows for immediate comparison between sides and can reveal subtle asymmetries that might otherwise go unnoticed. Gentle arm support by the examiner helps relax the pectoral muscles and provides better access to the axillary contents.
The examination should proceed systematically through each lymph node group, beginning with the anterior pectoral nodes along the medial border of the pectoralis minor muscle. The examining fingers should use gentle, circular motions with light to moderate pressure, avoiding excessive force that might cause discomfort or compress small nodes beyond detection. Normal lymph nodes typically feel soft, mobile, and measure less than 1 cm in diameter, whilst abnormal nodes may demonstrate increased size, firmness, or fixation to surrounding tissues.
Assessment of nodal characteristics requires careful attention to size, consistency, mobility, and tenderness, with each parameter providing valuable diagnostic information. Node size should be measured in centimetres using the largest dimension, with nodes exceeding 1 cm considered enlarged in most clinical contexts. Consistency varies from soft and rubbery in reactive nodes to firm or hard in malignant involvement, whilst mobility assessment determines whether nodes move freely or appear fixed to underlying structures.
The examination technique should include assessment of the surrounding skin and soft tissues, looking for signs of inflammation, infection, or other abnormalities that might explain nodal enlargement. Palpation of the supraclavicular and infraclavicular regions should be included as part of a comprehensive nodal examination, as involvement of these areas may indicate more extensive disease. Documentation of findings using standardised terminology ensures accurate communication between healthcare providers and facilitates appropriate follow-up care.
Patient education regarding self-examination techniques empowers women to monitor their own axillary lymph nodes between healthcare visits, potentially enabling earlier detection of significant changes. The self-examination should be performed monthly, preferably at the same time in the menstrual cycle to account for hormonal variations that might affect lymph node size. Women should be instructed to report any persistent enlargement lasting more than two weeks, particularly if accompanied by other concerning symptoms such as unexplained weight loss, night sweats, or persistent fatigue.